The diagram below can be used to determine enthalpy versus entropy of water and steam.
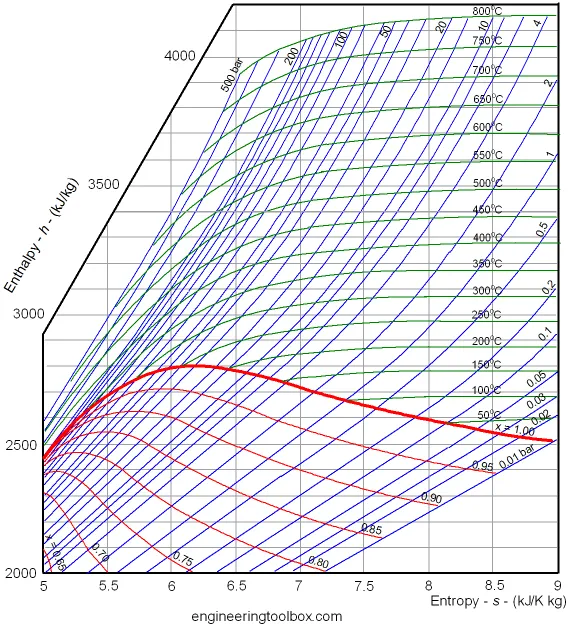
The Mollier diagram is useful when analyzing the performance of adiabatic steady-flow processes, such as flow in nozzles, diffusers, turbines and compressors.
See also Water – Enthalpy (H) and Entropy (S) for figures and tabulated values at varying temperatures
Dryness Fraction
The quantity of saturated vapor in unit mass of wet vapor, denoted by x, is referred to as the
dryness fraction, or quality,
of the vapor.
Convert to other Units
- 1 bar = 105 Pa (N/m2) = 0.1 N/mm2 = 10,197 kp/m2 = 10.20 m H2O = 0.9869 atm = 14.50 psi (lbf/in2) = 106 dyn/cm2 = 750 mmHg
- 1 kJ/kg = 0.4299 Btu/ lbm = 0.23884 kcal/kg
Example – Enthalpy
The enthalpy in saturated steam (x = 1, the bold red line in the diagram) with pressure 10 bar can be estimated to aprox.
2770 kJ/kg
The value can be verified by using the steam table.




