Phosphorus is known to form two types of halides PX3 and PX5. Of all the phosphorus halides formed, most common are phosphorus pentachloride, PCl5 and phosphorus trichloride, PCl3.
Phosphorus trichloride: It is a colourless oily liquid and highly toxic compound. The compound has triangular pyramidal shape in which phosphorus is sp3 hybridized.
Preparation:
• When dry chlorine is passed over heated white phosphorus, phosphorus trichloride is produced.
P4 + 6Cl2→4PCl3
• When thionyl chloride reacts with white phosphorus, phosphorus trichloride is produced.
P + 8SOCl2 → 4PCl3 + 4SO2 + 2S2Cl2
Chemical properties:
Phosphorus trichloride hydrolyses in presence of moisture.
PCl3 + 3H2O → H3PO3 + 3HCl
• It reacts with organic compounds containing –OH group to produce their chloro derivatives.
3C2H5OH + PCl3→3C2H5Cl + H3PO3
Phosphorus pentachloride: It is a yellowish white, water sensitive solid. It is soluble in carbon tetrachloride, carbon disulfide, benzene, diethyl ether. It has a trigonal bypyramidal structure in gaseous and liquid phases. In solid state, it exists as an ionic solid,[PCl4]+[PCl6]– in which the cation, [PCl4]+ is tetrahedral and the anion, [PCl6]– is octahedral.
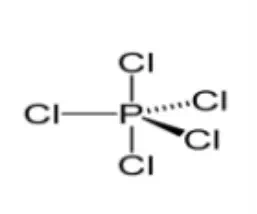
From the figure, we acknowledge the formation of three equatorial P-Cl bonds and two axial P-Cl bonds. Due to greater repulsion at axial positions in comparison to equatorial positions the two axial bonds are longer than equatorial bonds.
Preparation:
• When white phosphorus reacts with excess of dry chlorine, phosphorus pentachloride is produced.
P4 + 10Cl2→4PCl5
• It can also be prepared by the reaction of SO2Cl2 and phosphorus.
P4 + 10SO2Cl2→4PCl5+10SO2
Chemical properties:
• In moist air, phosphorus pentachloride hydrolyses to POCl3 which finally converts to phosphoric acid.
PCl5 + H2O→POCl3 + 2HCl
POCl3 + 3H2O→H3PO4 + 3HCl
• It sublimes on heating and further decomposes to phosphorus trichloride in case of stronger heating.
PCl5→PCl3 + Cl2
• It reacts with finely divided metals under the influence of heat to produce metal chlorides.
2Ag + PCl5→2AgCl + PCl3
• It reacts with organic compounds containing –OH group to produce their chloro derivatives.
C2H5OH + PCl5→C2H5Cl + POCl3 + HCl
Uses of phosphorus halides:
• It is used as a chlorinating agent.
• It is used as an intermediate in the manufacturing of phosphorus acid, chloroanhydrides and phosphoric acid derivatives.
• It is used as an intermediate for making organophosphorus pesticides, water treatment agents, lube oil and paint additives etc.




